HOPE for the liver – Hypothermic Oxygenated liver PErfusion
Hypothermic Oxygenated liver machine PErfusion (HOPE) – Better organ quality saves lifes
Concept of Hypothermic Oxygenated liver PErfusion (HOPE)
Principle
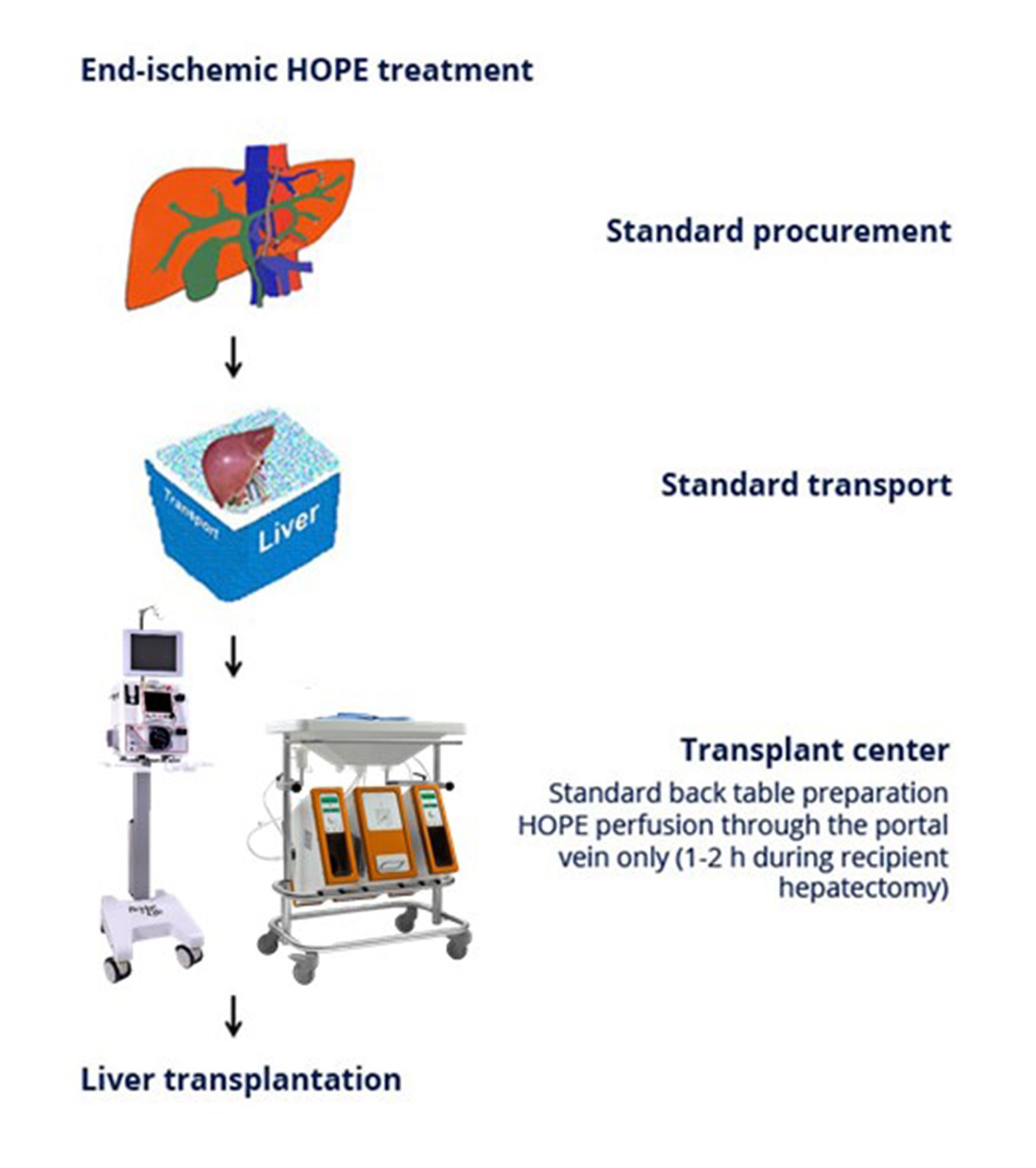
In an attempt to improve preservation of liver grafts, the concept of endischemic hypothermic oxygenated machine liver perfusion presents a simple, efficient and practical strategy to be used exclusively after organ transport at the site of organ transplantation.
Following standard back table preparation the provided disposable connects the liver with the perfusion machine (LiverAssist®, VitaSmart®), which is a simple approach, since perfusion, done while recipient hepatectomy, proceeds exclusively over the portal vein. Importantly, during 1 to 2 hours of perfusion portal pressure needs to be constantly monitored, aiming for values below 3 mmHg. Furthermore, the perfusate recquires full saturation with oxygen, while perfusate and liver temperature is maintained between 4 and 10 ° C. After hepatectomy of the recipient the perfused graft is disconnected and transplanted.
Protective Mechanism
The mechanisms of protection by hypothermic machine perfusion appear to be twofold.
First, oxygenation under hypothermic conditions protects from mitochondrial and nuclear injury by down regulation of mitochondrial activity before reperfusion. After one hour of perfusion, mitochondrial respiration appears significantly reduced in the presence of an oxygenated perfusate, suggesting that more than one h of HOPE treatment may not become necessary.
Second, cold perfusion itself, under low pressure conditions (≤ 3mmHg), cleans glycocalix and endothel, while preventing endothelial damage. Therefore, one hour HOPE in the transplant center, during recipient hepatectomy is enough, indiciating a very good liver function after reperfusion.
Publications
- Philipp Dutkowski et al. Hypothermic oxygenated machine perfusion (HOPE) down-regulates the immune response in a rat model of liver transplantation. Ann Surg 2014; 260(5): 931-938.
- Philipp Dutkowski, Pierre A. Clavien et al.First Comparison of Hypothermic Oxygenated Perfusion (HOPE) Versus Static Cold storage of Human DCD Liver Transplants – An International Matched Case Analysis.
Ann Surg 2015; 262: 764-771. - Philipp Dutkowski, Pierre A. Clavien et al. Outcomes of DCD liver transplantation using organs treated by hypothermic oxygenated perfusion before implantation. J Hepatol. 2019; 70: 50-57.
- Philipp Dutkowski, Pierre A. Clavien et al. Novel Real-time Prediction of Liver Graft Function During Hypothermic Oxygenated Machine Perfusion Before Liver Transplantation. Ann Surg. 2019; 270: 783-790.
- Philipp Dutkowski, Pierre A. Clavien et al. Hypothermic Oxygenated Liver Perfusion (HOPE) Prevents Tumor Recurrence in Liver Transplantation From Donation After Circulatory Death. Ann Surg. 2020; 272: 759-765.
- Mickaël Lesurtel, Philipp Dutkowski, Pierre A. Clavien et al. Hypothermic Oxygenated Perfusion Versus Normothermic Regional Perfusion in Liver Transplantation From Controlled Donation After Circulatory Death: First International Comparative Study. Ann Surg. 2020; 272: 751-758.
- van Rijn R, Schurink IJ, Porte RJ et al. Hypothermic Machine Perfusion in Liver Transplantation – A Randomized Trial. N Engl J Med. 2021 Feb 24. doi: 10.1056/NEJMoa2031532.
Human Trial – Study Design
Hypothermic Oxygenated liver PErfusion (HOPE)
To test machine liver perfusion against standard cold storage before liver transplantation.
The suggested study will demonstrate, for the first time, in a multicentric randomized trial the effect of a highly simplified endischemic perfusion technique in human donation after brain death (DBD) liver grafts in terms of post-transplant complications and costs. In case of convincing success in DBD livers, we postulate a high acceptance rate among transplant surgeons, since this approach can be easily applied in any center worldwide. The results are being expected by 2021.
Human trial